Natural-History-Conservation.com
Four-Day Fluid Preservation Course, November 2010, at The Horniman Public Museum & Public Park Trust London, UK.
 Schedule & course content:
Schedule & course content:
Day 1.
10.00 start. Introduction (incl. local logistics, fire-exits, risks, allergies).
Power-point 1: overview of course technology and histological effects of fluid preservation.
Questions and tea-break, discussion about the importance of fluid collections, fixation versus pseudo-fixation.
Compounding of sealant/s (gelatine, Acrifix, bitumen)
Glass cutting, grinding, drilling demonstrations and participation (demonstration & practical)
Rehydrations started (d & p)
Relevance of injecting fresh material with fixative
13.00 - 14.00. Lunch.
Preparation of jars (checking, grinding out blemishes – d & p)
Dehydration/ Hydration ladders start (d & p)
Thread mounting of specimens (d & p).
Celloidin mounting of snails to specimens and labels (d & p)
Check rehydrating specimens and fix + inject if they are ready, else leave (unheated) overnight (d & p).
Finish 1700.

 Day 2.
Day 2.
09.00 start. Power-point 2: narcotisation, historical sealants, pelagic (jellyfish) mounts, botanical preservatives.
Check of previous day’s work, analyse and correct problems. Move specimens in ladders (d & p)
Changing fluids in sealed jars (d & p).
Making glass needles and their use in specimen repair (d & p)
Celloidin specimen repair (d & p)
Releasing vacuum in Visijars (talk if none available)
Thread mounting of specimens (continuation) including jellyfish on acetate discs (d & p).
13.00 – 14.00 Lunch.
Move specimens in ladders (d & p)
Unsealing jars containing specimens requiring treatment (d & p)
Other types of sealant including historical sealants (d only)
Sealing of jars that are ready (d & p)
Finish 1700.
 Day 3.
Day 3.
09.00 start. Power-point 3: preservatives, lipids, copper wire staining
Checking of previous day’s seals (d & p).
Assessing problems: evaporation and dilution
What has happened if your Hirst is ‘off-colour’?
Dealing with lipids and other contaminants including fungal growth
Copper salts staining specimens, from being mounted on copper wire.
Detecting preserving fluids and auto-dilution problems: use of map pins and Dries van Dam’s ‘pills’ - Making your own specific gravity detector (d & p) Which preservatives should you use? (on hand-outs)
Problems of mixing fluids (exothermic due to binary azeotropy and leading to air bubble formation).
Buffering and pH levels (d & p).
13.00 - 14.00. Lunch.
How to deal with air bubbles, especially those trapped inside rehydrated specimens; (small portable vacuum pump required: d & p)
Stretching polypropylene rod (Bunsen burner/s required: d & p)
Topping up sealed jars, ‘corking’ and sealing.
Checking for problems and why they may have occurred.
 Day 4.
Day 4.
09.00 start. Power-point 4: Transparencies, labels, tubes in jars, jar types, storage areas. Maybe transporting and posting loan specimens if time allows.
Check of nearly-finished jars, are they leaking? If so, why, what has caused this – understanding how and why problems can occur and why deadlines can be difficult to meet! (d & p).
Dealing with transparencies – keeping fluids including glycerol, propylene glycol, methyl benzoate and turpentine and problems associated with these.
Sending fluid-preserved specimens by post or courier, worldwide policies changing year to year.
Which jars are best for your collection?
How to store such collections so that they need minimum monitoring and maintenance.
Labels and their problems, which paper/s to use. Internal or external labels?
13.00 - 14.00. Lunch.
Tubes in jars – how to seal, invert or not?
Storage areas. Plastination – ethics.
- 16.00 - Time overspill & Final questions.
- Depart

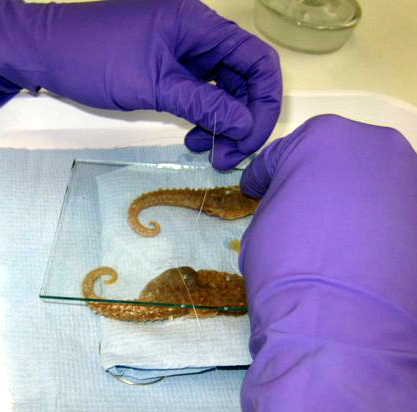
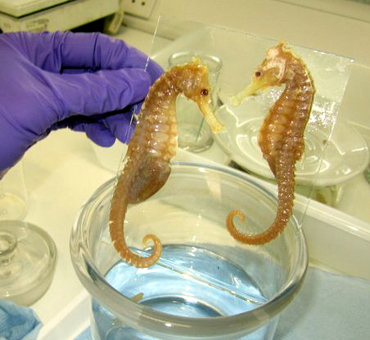

With thanks to John Simmons for the images.
An article from a recent SPNHC (Society for the Preservation of Natural History Collections) Newsletter about this course:

For more details about what we can do for you, or for a quote, please contact:
enquiries@natural-history-conservation.com
We are members of the United Kingdom Institute for Conservation of Historic and Artistic Works
Back to the home page
 Schedule & course content:
Schedule & course content: 
 Day 2.
Day 2. Day 3.
Day 3. Day 4.
Day 4.



